About
Pegaso MIE devices assist with airway clearance and mucus removal in a range of patients with respiratory insufficiency and are intended for home and hospital use. Suitable for both adults and children, the Pegaso E7C, Pegaso A-Cough and Pegaso Plus are used for IPV and cough assistance therapy. All devices are available for purchase, hire, or as part of an NDIS plan.
Conditions that may benefit from IPV and/or Assisted Cough include:
- Muscular Dystrophy (DMD, MD)
- Motor Neurons Disease (MND)
- Cerebral Palsy
- Spinal Muscular Atrophy (SMA)
- Spinal Cord Injury
- Traumatic Brain Injury (TBI)
- Polio
- Amyotrophic Lateral Sclerosis(ALS)
- Myasthenia Gravis
- Cystic Fibrosis – Also consider SIMEOX
- Chronic Obstructive Pulmonary Disease (COPD) – Also consider SIMEOX.
Pegaso E7C
Device Specifications:
The Pegaso E7C features two modes of operation, as well as a large colour touchscreen display for ease of use.
- 7 Different Modes
- Flow Aspire Mode (using Flow Aspire Adaptor)
- Passive expiratory flow acceleration for secretion clearance
- Mobilises mucus during natural breathing (no patient effort required)
- Protects collapsible airways by avoiding negative pressure
- Suitable for weak, ineffective, or non-cooperative patients.
- IPV Mode (Intrapulmonary Percussive Ventilation)
- Pressures up to 70cmH20
- Frequency 50-600 BPM
- IE Ratio 1:5 – 5:1 – (0.1 Increments)
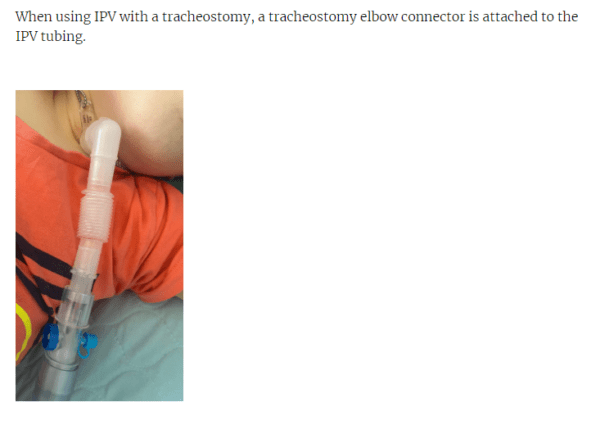
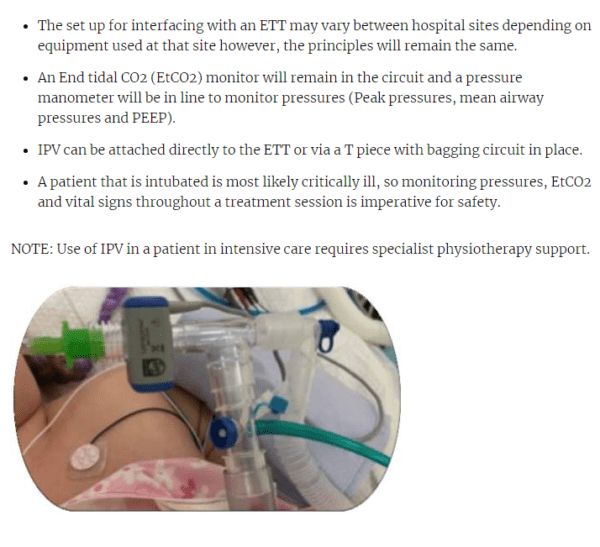
- In Percussor mode a humidifier can be attached and used with a mouthpiece, mask, ETT, or tracheostomy tube
- Cough Assist/Mi-E Mode(s)
- Automatic, Manual, Auto Adaptive, Air Stacking and Trainer
- Pressure up to 70cmH2O with extremely powerful flows for highly effective cough assist therapy
- Three different rise times
- Trigger sensitivity regulation (9 levels) for the auto-adaptive modes
- Programable air stacking up to 7 steps
- Display of inspired volumes and peak inspiratory flow
- Oscillatory function during cough assistance therapy on insufflation and exsufflation. Oscillatory Frequency and pressure can be set by the operator
- Live and recorded waveforms to help titrate Assisted Cough settings
- Mode or setting lock feature in the patient or carer menus
- Optional SpO2 monitoring directly from the display screen to help monitor lung perfusion and therapy efficacy
- Storage of 200 therapies
- USB, USB C and SD card for downloads and communication
- Weight 3.2kg
- Included internal battery with approximately 3-4 hours capacity
- HFCWO Belt (High Frequency Chest Wall Oscillation)
- Hands-free secretion clearance using the Pegaso E7C
- Delivers effective oscillation at 15 Hz (meets ≥13 Hz therapy threshold)
- Helps loosen and mobilise secretions while patients breathe normally
- Soft corset-style design for comfort and ease of use
- Suitable across a range of care settings and patient abilities
- Compatible with the Pegaso E7C.
- Optional foot pedal.
- TGA registered
- 2 Year Warranty.
Pegaso Plus Cough
A compact version of the Pegaso A-Cough, the Pegaso Plus Cough is designed for home use while still offering full functionality and an internal battery.
Device specifications:
- IPV mode
- Pressures up to 70cmH20,
- Frequency 10-600 BPM,
- IE Ratio 1:5 – 5:1
- Four different cough assistant modes:
- Automatic, Manual, AutoSync, EasyStart
- Pressure up to 70cmH2O with extremely powerful flows for highly effective cough assist therapy
- Three different rise times
- Trigger sensitivity regulation (9 levels) for the auto-adaptive modes (AutoSync, EasyStart)
- Display of inspired volumes and peak inspiratory flow
- Oscillatory function during cough assistance therapy
- Live and recorded waveforms to help titrate Assisted Cough settings
- Mode or setting lock feature in the patient or carer menus
- Optional SpO2 monitoring directly from the display screen to help monitor lung perfusion and therapy efficacy
- Storage of 200 therapies in internal memory
- Weight 3.2kg
- TGA registered
- Included internal battery with approximately 3 hours capacity
- Optional device trolley.
Pegaso A-Cough
Designed for hospital use, the Pegaso A-Cough is suitable for IPV and cough assistance therapy for neonates, paediatrics, and adults
Device specifications:
- IPV mode
- Pressures up to 70cmH20,
- Frequency 10-600 BPM,
- IE Ratio 1:5 – 5:1
- Four different cough assistant modes:
- Automatic, Manual, Auto Sync, EasyStart
- Pressure up to 70cmH20 with extremely powerful flows for highly effective cough assist therapy
- Three different rise times
- Display of inspired volumes and peak inspiratory flow
- Oscillatory function during cough assistance therapy
- Storage of 200 therapies in internal memory
- Weight 4.4kg
- TGA registered
- Optional SpO2 monitoring directly from the display screen to help monitor lung perfusion and therapy efficacy
- Optional device trolley.
Servicing
Service/performance verification battery change every 2 years.
To schedule a service for our device complete the form here and BMedical will be in touch to organise your service.
SERVICE AND WARRANTY FORM